The Caris+ App
Instant access to precision medicine.
Easily Access Your Patient’s Caris Case
The Caris+™ App provides convenient access to manage and review your Caris profiling cases. View current and past patient results whenever and wherever you are.
- View and search current and past cases
- Receive real-time case notifications
- Track testing progress
- Approve orders
- View interactive results and select clinical trial matches
- Submit cases for Caris Molecular Tumor Board review
- Share patient educational information
Real-Time Case Information
- Instant case access
- Case progress tracker
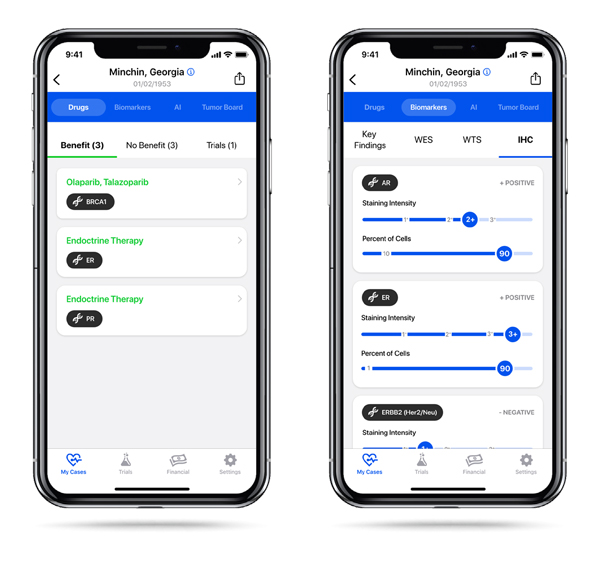
- Therapy associations
- Biomarker results
- One-touch access to online reference citations
- AI-driven results for Caris GPSai™ and FOLFIRSTai™
Clinical Trial Opportunities and Additional Features
- Real-time notifications for select patient-trial matches
- View trial inclusion/exclusion criteria
- Connect with clinical trial navigators regarding patient enrollment
- Request case review by Caris Molecular Tumor Board
- Send patients financial assistance information
- Access patient FAQs
Caris+ Frequently Asked Questions
Download the Caris+ App in the Apple App Store and the Google Play Store
Existing Caris MI Portal Users
Ordering physicians who have an active MI Portal account can log into the Caris+ App using their Portal email address and set a password by entering their name and email address. An email verification code will be sent to the email used for MI Portal. Users will then set up a password for Caris+, which can either be unique or the same password used for their existing MI Portal account.
Note: Setting a password in Caris+ will not change the MI Portal password.
New Users
New users who do not have a MI Portal account must select the New User option and complete the form to request an account. New accounts can take up to 48 business hours to be created. You will receive email confirmation when your MI Portal account has been created.
Once your MI Portal account has been created, users must complete signup for Caris+ by selecting Existing MI Portal User on the home screen and completing the contact and email verification steps.
When your account has been created, you will be able to use your email address and password to log into Caris+. Face or Touch ID options are available after the first sign in using the new password.
Once logged into the app, select the Settings icon at the bottom of your screen and select Account Information from the menu. You will need to re-enter your email address and password to make changes to your account.
Caris reports can be viewed and exported as a PDF by selecting the share icon in the top-right corner of the case details page. Users can share the report via email or save directly to their device.
Questions?
For questions or assistance about the Caris+ App, please contact customer support by email or phone.